What is it?
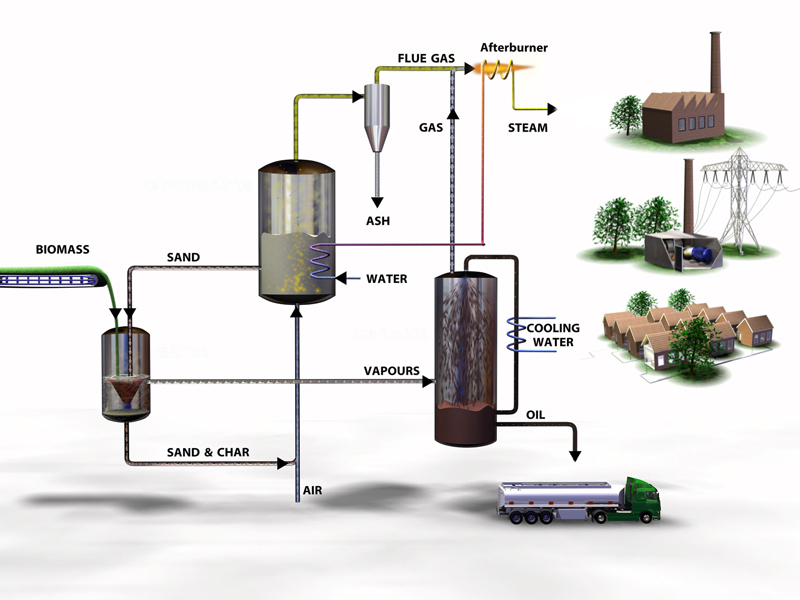
The technology that best proposed in converting the biomass (from the waste material) to the production of the biofuels is the PYROLYSIS TECHNOLOGY.
Why do you choose this technology? Why not others?
The reason why we choose this technology is simply because the ability of this technology in maintaining the good production the biofuels while at the same time does not have the safety concerns to the environment due to the lack of air emissions. The amount of greenhouse gas emissions in this process is significantly low until it only leads to minimal risk of health consequences .
Comparing the other technologies, anaerobic reaction might lead to the need to control emissions from burning biomass materials to prevent local air pollution. Meanwhile, the Hydrothermal liquefaction (HTL) requires high pressure and high temperature for the process, hence, it might lead to a high cost and possible safety concerns. Therefore, the pyrolysis technology is the best technology in biofuels production from biomass as well as the best green technology.
The technology that best proposed in converting the biomass (from the waste material) to the production of the biofuels is the PYROLYSIS TECHNOLOGY.
Why do you choose this technology? Why not others?
The reason why we choose this technology is simply because the ability of this technology in maintaining the good production the biofuels while at the same time does not have the safety concerns to the environment due to the lack of air emissions. The amount of greenhouse gas emissions in this process is significantly low until it only leads to minimal risk of health consequences .
Comparing the other technologies, anaerobic reaction might lead to the need to control emissions from burning biomass materials to prevent local air pollution. Meanwhile, the Hydrothermal liquefaction (HTL) requires high pressure and high temperature for the process, hence, it might lead to a high cost and possible safety concerns. Therefore, the pyrolysis technology is the best technology in biofuels production from biomass as well as the best green technology.
Is it a sustainable technology? (balanced between economy, social and environment)
A sustainable technology towards environment is a technology that will produce less pollutant (Gernand, 2008), which we usually defined as environment friendly technology. There was a bio-fuel concept: Carrying out open burning of plant materials will produce a lot of bad pollutants. Now, with the presence of pyrolysis, which is a process of heating the materials in a container without oxygen, most of the carbon released is known as ‘biochar’, which will actually act as an excellent soil additive (BioenergyWiki, n.d.). There are various environmental benefits including greenhouse gas stabilization by using fully sustainable biomass as feedstock in pyrolysis. Besides, the gas that is produced during pyrolysis can be processed into clean-burning fuel. Hence, it can be said that pyrolysis is a sustainable technology towards environment.
Economic sustainability must also be included in the criteria of a sustainable technology. Pyrolysis can be considered as a simple and low-cost technology capable of processing a wide variety of feedstock for example biomass from Tesco, Seri Iskandar to produce bio-oil. This plant involves the handling of biomass solids, a biomass pyrolysis reactor and a series of separation units to remove waste products from the syngas and isolate the bio-oil. The cost for the plant is a one-off cost, hence it will not cost much for the plant in long term. The cost of production depends heavily on the beginning in the field and ending at the product user. Since bio-oil is widely used in human’s daily life and an essential fuel for various activities, therefore, it will always be highly demanded. Overall, pyrolysis is considered an economically sustainable technology to convert waste product into bio-oil.
Societal impacts play a vital role in all the technologies. Since the feedstock for pyrolysis is the organic waste products from various sources, it provides waste collecting jobs for low-income people (BioenergyWiki, n.d.). It also provides opportunities for the skilled technicians as well as engineers to work hand in hand to run the reactor. Besides providing job opportunities, this technology helps to improve public health as it cleans up the waste materials around the communities by taking the waste materials as feedstock. Therefore, pyrolysis is defined as a sustainable technology as it has a balance between environment, economy and social.
Is there any catalyst involved? What type of catalyst? How much is required?
Catalyst always plays an important role in most of the chemical industry and researches. It is a substance that used to increases the rate of chemical reaction without being consumed or undergoing any changes at the end of reaction. Most of the chemical reactions require energy input to proceed and thus less energy will be required to complete the reaction if reaction is carried out in the presence of catalyst.
Fast pyrolysis is recognized worldwide as the one of the most efficient thermochemical technologies with the potential to convert cheap biomass into liquid fuel- bio oil. However bio-oil from fast pyrolysis contains large amount of oxygen, which cause many negative properties, such as low heating value, high viscosity, instability and also the limitation in application of bio-oil as transportation fuel (Liu & Wang, 2013). Therefore, converting biomass into transportation fuel with catalysts zeolite in fast pyrolysis reaction promotes catalytic cracking or bio-oil upgrading process which enhances the quality of bio-oil.
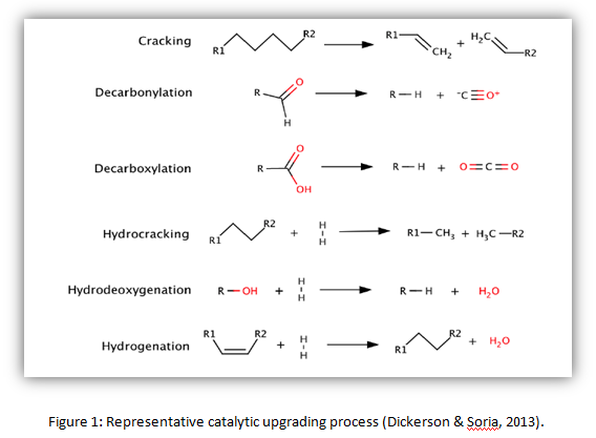
Zeolite catalyst is a type of highly deoxygenated and hydrocarbon-rich compounds, which able to exhibit catalytic activity in up to 50 percent of their volume. The main purpose of zeolite catalyst used in pyrolysis reaction is to convert oxygenated organic compounds into hydrocarbon and hence produce less-oxygenated bio-oil. The addition of catalysts to the pyrolysis system enhances catalytic cracking, which accomplishes deoxygenating through reactions such as decarboxylation, hydrocracking, hydrodeoxygenation, and hydrogenation. In catalytic upgrading process, properties of bio oil is being enhanced by removing oxygenated compounds via H2O and CO2, reducing molecular weight, and altering chemical structures to resemble those of petrochemical products. (Dickerson & Soria, 2013). Figure 1 below shows the catalytic upgrading process of bio-oil.
Generally, bio-oil is best upgraded by HZSM-5 or ZSM-5, as these zeolitic catalysts promote high yields of liquid products and propylene. Around 20 to 30 percent of aromatics are being yield in the presence of ZSM catalysts. Maximum hydrocarbon yields of 16 wt. %, including 3.5 wt. % toluene, were achieved with a nickel, cobalt, iron and palladium-substituted H-ZSM catalyst. CuO, mixed metal oxide catalysts and ZnO reduced of heavy fraction in the biooil yield. Pt on alumina showed the highest deoxygenating activity, reducing the oxygen concentration of the model bio-oil from 41.4 wt.% to 2.8 wt.% after upgrading (Dickerson & Soria, 2013). In particular attention is paid to the catalyst capabilities to suppress coke and gas formation, to reduce oxygen and modify its functionalities in the oil and to increase the yield of the hydrocarbon fraction. Researches shows that in case of using catalyst, the oxygen content decreased about 13-16 % and calorific value of bio-oil increased about 11-14 % compared with non-catalytic oil, which then important to make bio-oil become favorable to use as transportation fuel due to its instability and higher calorific value (Olay, 2014). Meanwhile, the upgraded oil will also comprise significant quantities of alkyl- substituted benzenes and cyclohexanes which consequently produce higher aromatic than non-catalytic bio-oil.
The usage of zeolite catalysts in fast pyrolysis can also slightly reduce the char product yield and favor the liquid product (bio-oil) yield about 9.1% compared to the non-catalyst pyrolysis reaction. However, the amount of zeolite catalyst used in the pyrolysis reaction is only depends on the amount of feedstock used. It was establish that any further increase of catalysts ratio used in the pyrolysis reaction will not results in any significant change in the amount of bio-oil yield.
The usage of zeolite catalysts in fast pyrolysis can also slightly reduce the char product yield and favor the liquid product (bio-oil) yield about 9.1% compared to the non-catalyst pyrolysis reaction. However, the amount of zeolite catalyst used in the pyrolysis reaction is only depends on the amount of feedstock used. It was establish that any further increase of catalysts ratio used in the pyrolysis reaction will not results in any significant change in the amount of bio-oil yield.
References
- BioenergyWiki (n.d.). Pyrolysis. Retrieved 27 July, 2015 from http://www.bioenergywiki.net/Pyrolysis#Products
- Dickerson, T. & Soria, J. (2013). Catalytic Fast Pyrolysis: A Review. Retrieved from 2 August, 2015 from http://library.certh.gr/libfiles/PDF/GEN-PAPYR-6177-CATALYTIC-by-DICKERSON-in-ENERGIES-V-6-PP-514-538-Y-2013.pdf
- Gernand, J. (May 9, 2008). Progressiprocity. Defining sustainable technology. Retrieved 27 July, 2015 from http://true-progress.com/defining-sustainable-technology-52.htm
- Liu, L. & Wang, H. (2013). Catalytic fast pyrolysis of lignocellulosic biomass. Retrieved from 2 August, 2015 from http://pubs.rsc.org/en/content/articlelanding/2014/cs/c3cs60414d#!divAbstract
- Onay, O. (2014.)Effects of Catalyst on Pyrolysis of Laurel (Laurus Nobilis L.) Seed in a Fixed Bed Tubular Reactor. Retrieved from 2 August, 2015 from http://www.aidic.it/cet/14/37/022.pdf
| 3_proposed_technology.pdf | |
| File Size: | 671 kb |
| File Type: | |